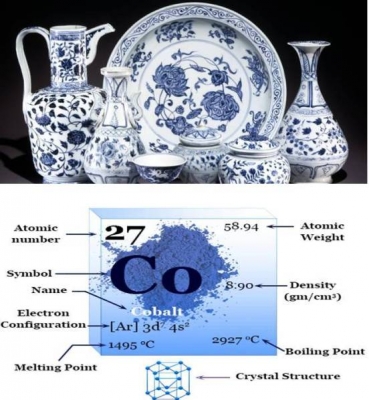
In ancient China, cobalt salts were used to decorate pottery with brilliant blue designs. Cobalt was discovered as an element in the 1730s. It was isolated by George Brandt, a Swedish chemist in 1735 for the first time.
The atomic number of cobalt is 27. In its pure form, cobalt is silvery-blue and brittle. Its properties are similar to iron and nickel. It can be made magnetic like iron. Because of this property, some high-powered magnets are made from alloys of cobalt and aluminium or nickel. Its man-made radioactive isotope, Cobalt-60, is used in cancer treatments.
Cobalt is also used in alloys to build parts of aircraft engines and in manufacturing alloys with corrosion/wear-resistant properties. It is widely used in making batteries and in electroplating. Cobalt is also an essential element for many living creatures. It is a component of vitamin B12.
In August 2014, a team of astrophysicists reported the presence of cobalt-56 in the supernova SN2014J which is an exploding star 11 million light-years from Earth!
Picture Credit : Google